The most recent iteration of Surviving Sepsis has been published in Critical Care Medicine and Intensive Care Medicine. The can be downloaded here (please click).
Below is a summary of the Guidelines:
A. Initial Resuscitation
1. Protocolized, quantitative resuscitation of patients with sepsis-induced tissue hypoperfusion (defined in this document as hypotension persisting after initial fluid challenge or blood lactate concentration ≥ 4 mmol/L).
Goals during the first 6 hrs of resuscitation:
a) Central venous pressure 8–12 mm Hg
b) Mean arterial pressure (MAP) ≥ 65 mm Hg c) Urine output ≥ 0.5 mL/kg/hr d) Central venous (superior vena cava) or mixed venous oxygen saturation 70% or 65%, respectively (grade 1C).
c). In patients with elevated lactate levels targeting resuscitation to normalize lactate (grade 2C).
B. Screening for Sepsis and Performance Improvement
1. Routine screening of potentially infected seriously ill patients for severe sepsis to allow earlier implementation of therapy (grade 1C).
2. Hospital–based performance improvement efforts in severe sepsis (UG).
C. Diagnosis
1. Cultures as clinically appropriate before antimicrobial therapy if no significant delay (> 45 mins) in the start of antimicrobial(s) (grade 1C). At least 2 sets of blood cultures (both aerobic and anaerobic bottles) be obtained before antimicrobial therapy with at least 1 drawn percutaneously and 1 drawn through each vascular access device, unless the device was recently (<48 hrs) inserted (grade 1C).
2. Use of the 1,3 beta-D-glucan assay (grade 2B), mannan and anti-mannan antibody assays (2C), if available, and invasive candidiasis is in differential diagnosis of cause of infection.
3. Imaging studies performed promptly to confirm a potential source of infection (UG).
D. Antimicrobial Therapy
1. Administration of effective intravenous antimicrobials within the first hour of recognition of septic shock (grade 1B) and severe sepsis without septic shock (grade 1C) as the goal of therapy.
2a. Initial empiric anti-infective therapy of one or more drugs that have activity against all likely pathogens (bacterial and/or fungal or viral) and that penetrate in adequate concentrations into tissues presumed to be the source of sepsis (grade 1B).
2b. Antimicrobial regimen should be reassessed daily for potential de-escalation (grade 1B).
3. Use of low procalcitonin levels or similar biomarkers (e.g. CRP) to assist the clinician in the discontinuation of empiric antibiotics in patients who initially appeared septic, but have no subsequent evidence of infection (grade 2C).
4a. Combination empirical therapy for neutropenic patients with severe sepsis (grade 2B) and for patients with difficult-to-treat, multidrug-resistant bacterial pathogens such as Acinetobacter and Pseudomonas spp. (grade 2B). For patients with severe infections
associated with respiratory failure and septic shock, combination therapy with an extended spectrum beta-lactam and either an aminoglycoside or a fluoroquinolone is for P. aeruginosa bacteremia (grade 2B). A combination of beta-lactam and macrolide for
patients with septic shock from bacteremic Streptococcus pneumoniae infections (grade 2B).
4b. Empiric combination therapy should not be administered for more than 3–5 days. De-escalation to the most appropriate single therapy should be performed as soon as the susceptibility profile is known (grade 2B).
5. Duration of therapy typically 7–10 days; longer courses may be appropriate in patients who have a slow clinical response, undrainable foci of infection, bacteremia with S. aureus; some fungal and viral infections or immunologic deficiencies, including neutropenia (grade 2C).
6. Antiviral therapy initiated as early as possible in patients with severe sepsis or septic shock of viral origin (grade 2C).
7. Antimicrobial agents should not be used in patients with severe inflammatory states determined to be of noninfectious cause (UG).
E. Source Control
1. A specific anatomical diagnosis of infection requiring consideration for emergent source control be sought and diagnosed or excluded as rapidly as possible, and intervention be undertaken for source control within the first 12 hr after the diagnosis is made, if feasible (grade 1C).
2. When infected peripancreatic necrosis is identified as a potential source of infection, definitive intervention is best delayed until adequate demarcation of viable and nonviable tissues has occurred (grade 2B).
3. When source control in a severely septic patient is required, the effective intervention associated with the least physiologic insult should be used (eg, percutaneous rather than surgical drainage of an abscess) (UG).
4. If intravascular access devices are a possible source of severe sepsis or septic shock, they should be removed promptly after other vascular access has been established (UG).
F. Infection Prevention
1a. Selective oral decontamination and selective digestive decontamination should be introduced and investigated as a method to reduce the incidence of ventilator-associated pneumonia; this infection control measure can then be instituted in health care settings and regions where this methodology is found to be effective (grade 2B).
1b. Oral chlorhexidine gluconate be used as a form of oropharyngeal decontamination to reduce the risk of ventilator-associated pneumonia in ICU patients with severe sepsis (grade 2B).
G. Fluid Therapy of Severe Sepsis
1. Crystalloids as the initial fluid of choice in the resuscitation of severe sepsis and septic shock (grade 1B).
2. Against the use of hydroxyethyl starches for fluid resuscitation of severe sepsis and septic shock (grade 1B).
3. Albumin in the fluid resuscitation of severe sepsis and septic shock when patients require substantial amounts of crystalloids (grade 2C).
4. Initial fluid challenge in patients with sepsis-induced tissue hypoperfusion with suspicion of hypovolemia to achieve a minimum of 30 mL/kg of crystalloids (a portion of this may be albumin equivalent). More rapid administration and greater amounts of fluid may be needed in some patients (grade 1C).
5. Fluid challenge technique be applied wherein fluid administration is continued as long as there is hemodynamic improvement either based on dynamic (eg, change in pulse pressure, stroke volume variation) or static (eg, arterial pressure, heart rate) variables (UG).
H. Vasopressors
1. Vasopressor therapy initially to target a mean arterial pressure (MAP) of 65 mm Hg (grade 1C).
2. Norepinephrine as the first choice vasopressor (grade 1B).
3. Epinephrine (added to and potentially substituted for norepinephrine) when an additional agent is needed to maintain adequate blood pressure (grade 2B).
4. Vasopressin 0.03 units/minute can be added to norepinephrine (NE) with intent of either raising MAP or decreasing NE dosage (UG).
5. Low dose vasopressin is not recommended as the single initial vasopressor for treatment of sepsis-induced hypotension and vasopressin doses higher than 0.03-0.04 units/minute should be reserved for salvage therapy (failure to achieve adequate MAP with other vasopressor agents) (UG).
6. Dopamine as an alternative vasopressor agent to norepinephrine only in highly selected patients (eg, patients with low risk of tachyarrhythmias and absolute or relative bradycardia) (grade 2C).
7. Phenylephrine is not recommended in the treatment of septic shock except in circumstances where (a) norepinephrine is associated with serious arrhythmias,
(b) cardiac output is known to be high and blood pressure persistently low or (c) as salvage therapy when combined inotrope/vasopressor drugs and low dose vasopressin have failed to achieve MAP target (grade 1C).
8. Low-dose dopamine should not be used for renal protection (grade 1A).
9. All patients requiring vasopressors have an arterial catheter placed as soon as practical if resources are available (UG).
I. Inotropic Therapy
1. A trial of dobutamine infusion up to 20 micrograms/kg/min be administered or added to vasopressor (if in use) in the presence of (a) myocardial dysfunction as suggested by elevated cardiac filling pressures and low cardiac output, or (b) ongoing signs of
hypoperfusion, despite achieving adequate intravascular volume and adequate MAP (grade 1C).
2. Not using a strategy to increase cardiac index to predetermined supranormal levels (grade 1B).
J. Corticosteroids
1. Not using intravenous hydrocortisone to treat adult septic shock patients if adequate fluid resuscitation and vasopressor therapy are able to restore hemodynamic stability (see goals for Initial Resuscitation). In case this is not achievable, we suggest intravenous hydrocortisone alone at a dose of 200 mg per day (grade 2C).
2. Not using the ACTH stimulation test to identify adults with septic shock who should receive hydrocortisone (grade 2B).
3. In treated patients hydrocortisone tapered when vasopressors are no longer required (grade 2D).
4. Corticosteroids not be administered for the treatment of sepsis in the absence of shock (grade 1D).
5. When hydrocortisone is given, use continuous flow (grade 2D).
K. Blood Product Administration
1. Once tissue hypoperfusion has resolved and in the absence of extenuating circumstances, such as myocardial ischemia, severe hypoxemia, acute hemorrhage, or ischemic heart disease, we recommend that red blood cell transfusion occur only when hemoglobin concentration decreases to <7.0 g/dL to target a hemoglobin concentration of 7.0 –9.0 g/dL in adults (grade 1B).
2. Not using erythropoietin as a specific treatment of anemia associated with severe sepsis (grade 1B).
3. Fresh frozen plasma not be used to correct laboratory clotting abnormalities in the absence of bleeding or planned invasive procedures (grade 2D).
4. Not using antithrombin for the treatment of severe sepsis and septic shock (grade 1B).
5. In patients with severe sepsis, administer platelets prophylactically when counts are <10,000/mm3 (10 x 109/L) in the absence of apparent bleeding. We suggest prophylactic platelet transfusion when counts are < 20,000/mm3 (20 x 109/L) if the patient has a significant risk of bleeding. Higher platelet counts (≥50,000/mm3 [50 x 109/L]) are advised for active bleeding, surgery, or invasive procedures (grade 2D).
L. Immunoglobulins
1. Not using intravenous immunoglobulins in adult patients with severe sepsis or septic shock (grade 2B).
M. Selenium
1. Not using intravenous selenium for the treatment of severe sepsis (grade 2C).
N. History of Recommendations Regarding Use of rhAPC
A history of the evolution of SSC recommendations as to rhAPC (no longer available) is provided.
O. Mechanical Ventilation of Sepsis-Induced ARDS
1. Target a tidal volume of 6 mL/kg predicted body weight in patients with sepsis-induced ARDS (grade 1A vs. 12 mL/kg).
2. Plateau pressures be measured in patients with ARDS and initial upper limit goal for plateau pressures in a passively inflated lung be ≤30 cm H2O (grade 1B).
3. Positive end-expiratory pressure (PEEP) be applied to avoid alveolar collapse at end expiration (atelectotrauma) (grade 1B).
4. Strategies based on higher rather than lower levels of PEEP be used for patients with sepsis-induced moderate or severe ARDS (grade 2C).
5. Recruitment maneuvers be used in sepsis patients with severe refractory hypoxemia (grade 2C).
6. Prone positioning be used in sepsis-induced ARDS patients with a Pao2/Fio2 ratio ≤100 mm Hg in facilities that have experience with such practices (grade 2B).
7. That mechanically ventilated sepsis patients be maintained with the head of the bed elevated to 30-45 degrees to limit aspiration risk and to prevent the development of ventilator-associated pneumonia (grade 1B).
8. That noninvasive mask ventilation (NIV) be used in that minority of sepsis-induced ARDS patients in whom the benefits of NIV have been carefully considered and are thought to outweigh the risks (grade 2B).
9. That a weaning protocol be in place and that mechanically ventilated patients with severe sepsis undergo spontaneous breathing trials regularly to evaluate the ability to discontinue mechanical ventilation when they satisfy the following criteria:
a) arousable; b) hemodynamically stable (without vasopressor agents); c) no new potentially serious conditions; d) low ventilatory and end-expiratory pressure requirements; and e) low Fio2 requirements which can be met safely delivered with a face mask or nasal cannula. If the spontaneous breathing trial is successful, consideration should be given for extubation (grade 1A).
10. Against the routine use of the pulmonary artery catheter for patients with sepsis induced ARDS (grade 1A).
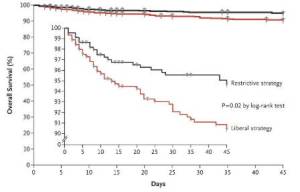
11. A conservative rather than liberal fluid strategy for patients with established sepsis-induced ARDS who do not have evidence of tissue hypoperfusion (grade 1C).
12. In the absence of specific indications such as bronchospasm, not using beta 2-agonists for treatment of sepsis-induced ARDS (grade 1B).
P. Sedation, Analgesia, and Neuromuscular Blockade in Sepsis
1. Continuous or intermittent sedation be minimized in mechanically ventilated sepsis patients, targeting specific titration endpoints (grade 1B).
2. Neuromuscular blocking agents (NMBAs) be avoided if possible in the septic patient without ARDS due to the risk of prolonged neuromuscular blockade following discontinuation. If NMBAs must be maintained, either intermittent bolus as
required or continuous infusion with train-of-four monitoring of the depth of blockade should be used (grade 1C). 3. A short course of NMBA of not greater than 48 hours for patients with early sepsis-induced ARDS and a Pao2/Fio2 < 150 mm Hg (grade 2C).
Q. Glucose Control
1. A protocolized approach to blood glucose management in ICU patients with severe sepsis commencing insulin dosing when 2 consecutive blood glucose levels are >180 mg/dL. This protocolized approach should target an upper blood glucose ≤180 mg/dL rather than an upper target blood glucose ≤ 110 mg/dL (grade 1A).
2. Blood glucose values be monitored every 1–2 hrs until glucose values and insulin infusion rates are stable and then every 4 hrs thereafter (grade 1C).
3. Glucose levels obtained with point-of-care testing of capillary blood be interpreted with caution, as such measurements may not accurately estimate arterial blood or plasma glucose values (UG).
R. Renal Replacement Therapy
1. Continuous renal replacement therapies and intermittent hemodialysis are equivalent in patients with severe sepsis and acute renal failure (grade 2B).
2. Use continuous therapies to facilitate management of fluid balance in hemodynamically unstable septic patients (grade 2D).
S. Sodium Bicarbonate Therapy
1. Not using sodium bicarbonate therapy for the purpose of improving hemodynamics or reducing vasopressor requirements in patients with hypoperfusion-induced lactic acidemia with pH ≥7.15 (grade 2B).
T. Deep Vein Thrombosis Prophylaxis
1. Patients with severe sepsis receive daily pharmacoprophylaxis against venous thromboembolism (VTE) (grade 1B). This should be accomplished with daily subcutaneous low-molecular weight heparin (LMWH) (grade 1B versus twice daily UFH, grade 2C versus three times daily UFH). If creatinine clearance is <30 mL/min, use dalteparin (grade 1A) or another form of LMWH that has a low degree of renal metabolism (grade 2C) or UFH (grade 1A).
2. Patients with severe sepsis be treated with a combination of pharmacologic therapy and intermittent pneumatic compression devices whenever possible (grade 2C).
3. Septic patients who have a contraindication for heparin use (eg, thrombocytopenia, severe coagulopathy, active bleeding, recent intracerebral hemorrhage) not receive pharmacoprophylaxis (grade 1B), but receive mechanical prophylactic treatment, such
as graduated compression stockings or intermittent compression devices (grade 2C), unless contraindicated. When the risk decreases start pharmacoprophylaxis (grade 2C).
U. Stress Ulcer Prophylaxis
1. Stress ulcer prophylaxis using H2 blocker or proton pump inhibitor be given to patients with severe sepsis/septic shock who have bleeding risk factors (grade 1B).
2. When stress ulcer prophylaxis is used, proton pump inhibitors rather than H2RA (grade 2D)
3. Patients without risk factors do not receive prophylaxis (grade 2B).
V. Nutrition
1. Administer oral or enteral (if necessary) feedings, as tolerated, rather than either complete fasting or provision of only intravenous glucose within the first 48 hours after a diagnosis of severe sepsis/septic shock (grade 2C).
2. Avoid mandatory full caloric feeding in the first week but rather suggest low dose feeding (eg, up to 500 calories per day), advancing only as tolerated (grade 2B).
3. Use intravenous glucose and enteral nutrition rather than total parenteral nutrition (TPN) alone or parenteral nutrition in conjunction with enteral feeding in the first 7 days after a diagnosis of severe sepsis/septic shock (grade 2B).
4. Use nutrition with no specific immunomodulating supplementation rather than nutrition providing specific immunomodulating supplementation in patients with severe sepsis (grade 2C).
W. Setting Goals of Care
1. Discuss goals of care and prognosis with patients and families (grade 1B).
2. Incorporate goals of care into treatment and end-of-life care planning, utilizing palliative care principles where appropriate (grade 1B).
3. Address goals of care as early as feasible, but no later than within 72 hours of ICU admission (grade 2C).
Source: Dellinger RP, Levy MM, Rhodes A, et al: Surviving Sepsis Campaign: International guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013; 41:580-637